Which bond is stronger ionic or polar covalent?
Which bond is stronger ionic or polar covalent?
Ionic Bonds are stronger than covalent bonds because the electronegativity difference between the two elements is much greater than that of two elements in a covalent bond. Covalent bonds allow the electrons to be shared between the two elements and will often favor one element over the other depending on polarity.
Are polar covalent bonds ionic?
Bonds that are partly ionic are called polar covalent bonds. Nonpolar covalent bonds, with equal sharing of the bond electrons, arise when the electronegativities of the two atoms are equal.
Which bond is the strongest bond?
covalent bond
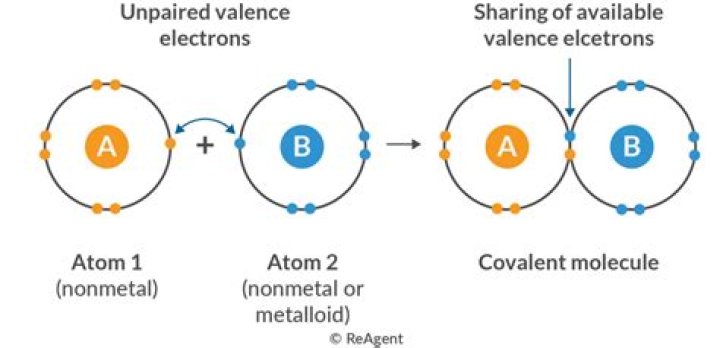
In chemistry, covalent bond is the strongest bond. In such bonding, each of two atoms shares electrons that binds them together. For example, water molecules are bonded together where both hydrogen atoms and oxygen atoms share electrons to form a covalent bond.
What is the weakest type of bond?
ionic bond
The ionic bond is generally the weakest of the true chemical bonds that bind atoms to atoms.
What is the difference between polar covalent and nonpolar covalent?
A covalent bond that has an unequal sharing of electrons, as in part (b) of Figure 4.4. 1, is called a polar covalent bond. A covalent bond that has an equal sharing of electrons (part (a) of Figure 4.4. 1) is called a nonpolar covalent bond.
Which type of bond is the weakest?
Why is covalent stronger than ionic?
Because of the close sharing of pairs of electrons (one electron from each of two atoms), covalent bonds are stronger than ionic bonds.
What are polar covalent bonds?
A polar covalent bond exists when atoms with different electronegativities share electrons in a covalent bond. The unequal sharing of the bonding pair results in a partial negative charge on the chlorine atom and a partial positive charge on the hydrogen atom.
What are the 4 types of bonds?
The properties of a solid can usually be predicted from the valence and bonding preferences of its constituent atoms. Four main bonding types are discussed here: ionic, covalent, metallic, and molecular.
What is the difference between ionic nonpolar covalent and polar covalent bonds?
An ionic bond is when there is a transfer of one or more electrons from a metal onto a non-metal. The general rule of thumb is that if the difference in electronegativity is greater than 2, then it is ionic. A covalent bond is the sharing of electrons, nonpolar is equal sharing and polar is unequal sharing.
What is the difference between covalent bonds and polar covalent?
Covalent bonds form when electrons are shared between atoms and are attracted by the nuclei of both atoms. In pure covalent bonds, the electrons are shared equally. In polar covalent bonds, the electrons are shared unequally, as one atom exerts a stronger force of attraction on the electrons than the other.