What is the equation for energy levels?
What is the equation for energy levels?
In the next section, let us look at the formula used to calculate the energy of the electron in the nth energy level….Summary.
| Value of the Atomic Radius | r(n)=n2×r(1) |
|---|---|
| The formula for defining energy level | E=E0n2 |
What is Bohr’s equation for energy levels?
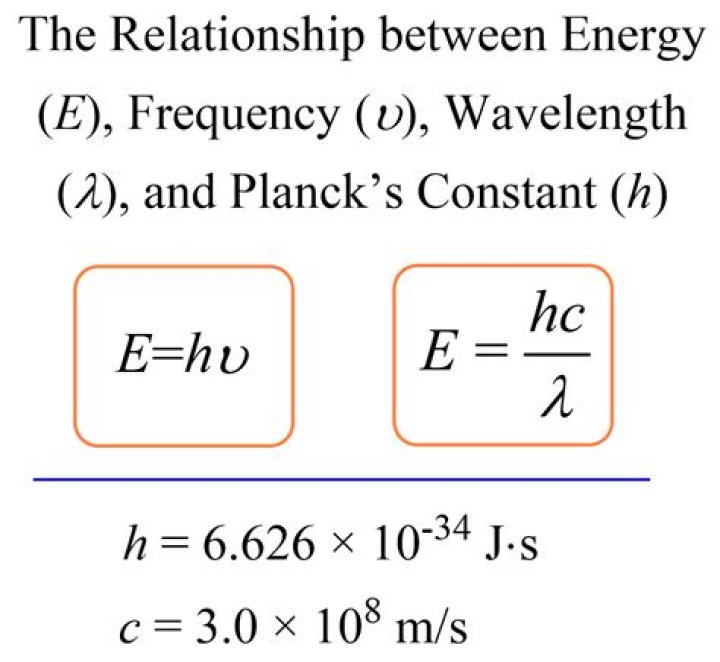
Bohr correctly proposed that the energy and radii of the orbits of electrons in atoms are quantized, with energy for transitions between orbits given by ∆E = hf = Ei − Ef, where ∆E is the change in energy between the initial and final orbits and hf is the energy of an absorbed or emitted photon.
What are the 4 energy levels?
| Orbitals and Electron Capacity of the First Four Principle Energy Levels | ||
|---|---|---|
| Principle energy level (n) | Type of sublevel | Number of orbitals per level(n2) |
| 4 | s | 16 |
| p | ||
| d |
What is energy level diagram?
The energy level diagram is used to represent the energy states available in each atom. When an electron is in an energy state, it emits nor absorbs radiation. A photon is emitted or absorbed when an electron transitions from one energy state to another.
What is ze 2 r2?
2. From Coulomb’s Law, the force of attraction between the electron and the nucleus is Ze2/r2, where Z is the charge on the nucleus (the atoms atomic number) and e is the electron’s elementary charge.
What’s an energy level diagram?
An energy level diagram shows whether a reaction is exothermic or endothermic . It shows the energy in the reactants and products , and the difference in energy between them.
What is energy level diagram in physics?
What are the energy levels of electron?
What Are Energy Levels? Energy levels (also called electron shells) are fixed distances from the nucleus of an atom where electrons may be found. Electrons are tiny, negatively charged particles in an atom that move around the positive nucleus at the center. Energy levels are a little like the steps of a staircase.
How many energy levels are there?
7 energy levels
Electron Shells, Subshells & Atomic Orbitals The highest energy level number (1 through 7) for the electrons in an atom corresponds to the period (or row) in the periodic table to which that atom belongs. Because there are 7 periods in the table, there are 7 energy levels.
What does the Bohr model do?
The Bohr model shows the atom as a central nucleus containing protons and neutrons with the electrons in circular orbitals at specific distances from the nucleus (Figure 1). These orbits form electron shells or energy levels, which are a way of visualizing the number of electrons in the various shells.
How do you write a mo diagram?
FUNDAMENTAL STEPS IN DERIVING MO DIAGRAMS
- Find the valence electron configuration of each atom in the molecule.
- Decide if the molecule is homonuclear of heteronuclear.
- Fill molecular orbitals using energy and bonding properties of the overlapping atomic orbitals.
- Use the diagram to predict properties of the molecule.
What is an energy level diagram?
Energy level diagrams are a means of analyzing the energies electrons can accept and release as they transition from one accepted orbital to another.
What is the value of energy emitted for a specific transition?
The value of the energy emitted for a specific transition is given by the equation [latex]hv=\\Delta E=(\\frac{1}{n_{low}^2}-{\\frac{1}{n_{high}^2}})13.6\\,eV[/latex] The formula for defining energy level
What is the degeneracy of the j = 1 energy level?
The degeneracy of the J = 1 energy level is 3 because there are three states with the energy 2ℏ2 2I. J = 2: The next energy level is for J = 2. The energy is 6ℏ2 2I, and there are five states with this energy corresponding to mJ = + 2, + 1, 0, ‑ 1, ‑ 2. The energy level degeneracy is five.
How do you calculate the energy level of an atom?
Using the Bohr Model, the energy levels (in electron volts, eV) are calculated with the formula: where Z is the atomic number and n is the energy level.