What is the difference between a monomer & polymer?
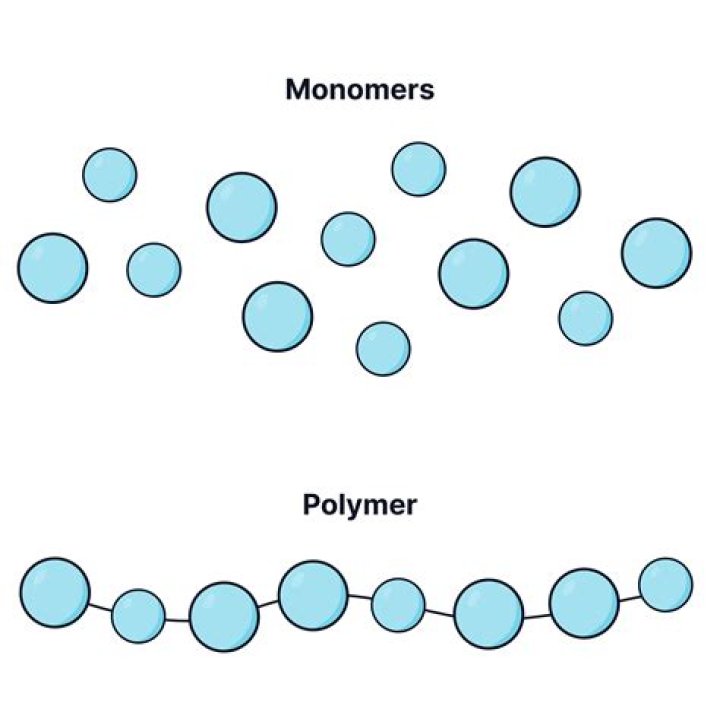
What is the difference between a monomer & polymer?
Monomers are small molecules, mostly organic, that can join with other similar molecules to form very large molecules, or polymers. Polymers are a class of synthetic substances composed of multiples of simpler units called monomers. Polymers are chains with an unspecified number of monomeric units.
What are natural polymers?
Natural Polymers are those substances which are obtained naturally. These polymers are formed either by the process of addition polymerization or condensation polymerization. Polymers are extensively found in nature. Our body too is made up of many natural polymers like nucleic acids, proteins, etc.
Why do polymers behave differently than monomers?
One major difference between polymers and small molecules (monomers) are the characteristics that polymers exhibit. Polymers normally have higher viscosities, higher boiling points and can show improved mechanical strength over small molecules (monomers).
Is polymer bigger than monomers?
A monomer is a single atom, small molecule, or molecular fragment that, when bonded together with identical and similar types of monomers, form a larger, macromolecule known as a polymer. Because there are so many different monomers that can be combined in numerous ways, there are many kinds of plastics.
What is the difference between a monomer and a polymer of a macromolecule?
These units are called repeating units. These repeating units represent the monomers from which the polymer is made. The main difference between polymer and macromolecule is that polymers contain repeating units that represent the monomers whereas not all macromolecules have a monomer in their structure.
What are the 4 natural polymers?
Natural polymers include:
- Proteins, such as hair, nails, tortoiseshell.
- Cellulose in paper and trees.
- Starches in plants such as potatoes and maize.
- DNA.
- Pitch (also known as bitumen or tar)
- Wool (a protein made by animals)
- Silk (a protein made by insects)
- Natural rubber and lacquer (proteins from trees)
How do I identify a monomer?
The simplest way to identify a monomer is to look at its structure. It always contains different combinations of atoms that together form a unique molecule having a molecular formula in accordance with the general formula of that class. For example, the general formula for monomers of carbohydrates is (CH2O)x.
What is the meaning of depolymerization?
Definition of depolymerize transitive verb. : to decompose (macromolecules) into simpler compounds (such as monomers)
Why different polymers have different properties?
Why do different polymers have different properties? Answer: They have different chemical compositions (different monomer units), different structures, different ways of being fabricated, etc.
What are monomers made of?
Monomers are small molecules that can combine to form larger molecules called polymers. Amino acids make up proteins. Monosaccharides such as glucose make up polysaccharides like starches.
What is the difference between monomers and polysaccharides?
Explanation: Monomers are small molecules that can combine to form larger molecules called polymers. Amino acids make up proteins. Monosaccharides such as glucose make up polysaccharides like starches. Serena D. · 1 · Mar 25 2018
How do monomers link to form branched polymers?
Normally, there are two functional groups in both sides of the monomer, so it can form linear chains by binding to other molecules from both sides. If there are several functional groups, monomers can link to form branched polymers. For example, glucose is the common monomer of carbohydrates with –OH functional groups.
What is the difference between fatty acids and monomers?
Monomers are smaller molecules, and when bonded together, make up polymers . -Fatty acids are the monomers for lipids , for example, and regardless of how they are bonded (as a saturated or unsaturated fat, for example), they will form lipids.